This website uses cookies as well as similar tools and technologies to understand visitors’ experiences. By continuing to use this website, you consent to Columbia University’s usage of cookies and similar technologies, in accordance with the Columbia University Website Cookie Notice.
Energy Explained
Insights from the Center on Global Energy Policy
Demystifying Electrolyzer Production Costs
This Energy Explained post represents the research and views of the author. It does not necessarily represent the views of the Center on Global Energy Policy. The piece may be subject to further revision. Contributions to SIPA for the benefit of CGEP are general use gifts, which gives the Center discretion in how it allocates these funds. Rare cases of sponsored projects are clearly indicated.
For a full list of financial supporters of the Center on Global Energy Policy at Columbia University SIPA, please visit our website at Our Partners. See below a list of members that are currently in CGEP’s Visionary Circle. This list is updated periodically.
Renewable hydrogen must achieve dramatic cost reductions[1] if it is to compete with existing fossil-fuel-based hydrogen as well as enable new uses. Electrolyzer costs are the biggest cost component of renewable hydrogen production, along with electricity. This article breaks down the primary components of renewable hydrogen production costs at a time when national hydrogen policies are targeting a reduction of overall electrolyzer costs.[2]
Sweeping statements about the relative costs of renewable hydrogen production methods and potential improvements can be tenuous. Not only are there many kinds of electrolyzers, but complexity in their design, set-up, and operation leads to variations in production costs. The incredible pace of industry innovation behind closed doors and lack of public, up-to-date data also exacerbate this challenge. Policymakers focused on driving down renewable hydrogen costs should consider upfront capital expenditure, efficiency and electricity costs, stack degradation and replacement rates, the impact of a module’s size, and potential economies of scale in manufacturing, as well as how well each electrolyzer types integrates for operation with intermittent renewables and specific industrial applications. Crucially, improvements in one parameter may negatively impact others.
Moreover, the cost ramifications vary—both in terms of the cost of the electrolyzer stack itself and the effectiveness and design of the resulting hydrogen production system—depending on the electrolyzer type.[3] The four most promising commercial electrolyzer types today are (Figure 1):
- Alkaline electrolyzer: Industry incumbent with decades of operating experience; low cost but only pressurized electrolyzers can ramp up and down to match renewable electricity
- Anion exchange electrolyzer (AEM): Potentially competitive with alkaline on cost; can ramp up with renewables but faces skepticism over degradation rates
- Proton exchange membrane electrolyzer (PEM): Recently commercialized; ideal for quick ramping up with renewables, but high material costs
- Solid oxide electrolyzer (SOEC): High-temperature electrolyzer with the highest efficiency potential, well-suited for hard-to-abate industrial processes with waste heat[4]; can ramp up and down with renewables, but offers lower durability
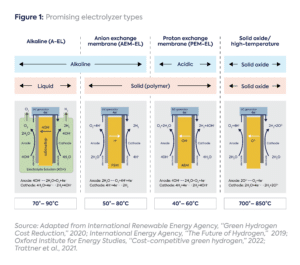
Below the authors discuss the complex factors influencing the costs of these electrolyzers and how they ultimately impact the cost of renewable hydrogen.
Upfront Capital Costs
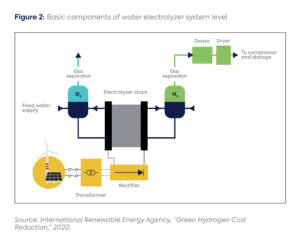
At the system level, one needs to distinguish between an electrolyzer’s stack—where the hydrogen production takes place—and the balance of plant (BoP), which refers to the miscellaneous equipment surrounding the stack, including for cooling, compression, purification, power electronics, and water treatment (Figure 2).[5] The stack(s) within a unit are sometimes called a module.
Traditional alkaline electrolyzers require the lowest upfront capital expenditure because they are built from low-cost steel or nickel-alloy-plated input materials.[6] However, the corrosiveness and complexity involved in controlling the concentration of potassium hydroxide creates reliability issues that add costs.[7] The upfront costs of the more novel PEM electrolyzers are higher, because only distinct metals can withstand both the corrosive acidic conditions and high applied voltage for high rates of hydrogen production.[8] PEM requires expensive platinum-group metals, notably iridium, limiting the level of cost reduction achievable.[9] In contrast, SOEC relies on abundant raw materials such as nickel, zirconia, and steel.[10] In its pre-commercial state, its upfront capex is higher than alkaline or PEM—with challenges including the high complexity of its manufacturing process—but, given its early development stage, greater cost reductions are expected.[11] Finally, some manufacturers are betting on the AEM electrolyzer for its cheap materials, expecting performance comparable to PEM at a lower cost.[12]
The wide range in electrolyzer capital costs (including BoP) speaks to the uncertainty surrounding numbers that are closely guarded by industry, and that quickly become obsolete by innovation (Table 1). Academia, too, appears to rely on data that has been circulating for some time: for instance, a 2022 Oxford Institute for Energy Studies paper cites Böhm et al. (2019), which cites Fraunhofer (2018) for its 2017 industry surveys.[13] The International Energy Agency’s 2019 numbers are similarly derived from sources published from 2018 to 2014, including the Fraunhofer study.[14] Furthermore, the combination of increased deployment and scale may make these numbers quickly out of date.
Efficiency Considerations Around Electricity Use
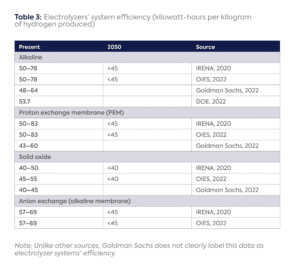
As the number of full load hours increases,[15] the cost impact of an electrolyzer’s initial capital expenditure declines and electricity becomes the primary cost component for renewable hydrogen production.[16] Efficiency ranges[17] vary significantly among electrolyzer types, with SOEC showing the highest efficiency (Table 2). Efficiency is also expressed as the electricity consumption to produce one kilogram of hydrogen at the system level, where lower numbers signify greater efficiency (Table 3).
The type of electrolyzer used may depend on the availability and cost of local electricity. A common industry view is that a higher efficiency, higher capex electrolyzer (such as SOEC) could be used in locations where renewable electricity is scarce, whereas a lower capex, lower efficiency electrolyzer (such as alkaline) could be used in places with plentiful renewable resources more focused on minimizing upfront capex costs.[18]
The variation in efficiency data suggests that considerable uncertainty persists today that could potentially be reduced through operating data from the first renewable hydrogen installations. But that data in turn will depend on the operations of electrolyzers; in most cases, there is considerable scope for efficiency improvement.
Stack Degradation Rates and Replacement Costs
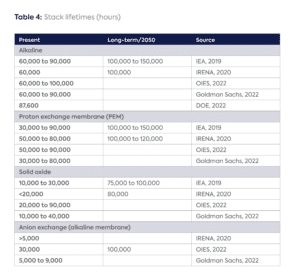
To complicate cost assessment further, the core of the electrolyzer (stack) will degrade over time depending on how the electrolyzer is assembled and operated, requiring replacement before the end of the project life. Factors affecting the lifetime of a stack include the operating current density,[19] temperature, pressure, the extent to which the electrolyzer ramps up and down to match renewable electricity, as well as sources of impurities and corrosion, such as water impurities or gas permeation across the membrane.[20] This cost could be financed either as an additional capital or operating cost in the year the stack requires replacement,[21] and would vary on a case-by-case basis.
Stack lifetimes are currently estimated to fall in the range of 3.5 to 11.5 years, while stacks account for between 19 and 60 percent of upfront electrolyzer capital costs,[22] depending on electrolyzer type. Alkaline and PEM electrolyzers today have longer stack lifetimes than SOEC and AEM, but their stack costs are also higher relative to the system costs. Stack costs of SOEC and AEM electrolyzers are estimated to be as low as 30 percent and 19 percent of total system costs, respectively.[23] The share of stack costs may increase in parallel with module size as better cost improvements are achieved with the BoP.[24]
Future Economies of Scale
Achieving future cost reductions also depends on achieving economies of scale, both by increasing the size of the module and the size of the manufacturing plant. Increasing the size of the module enables cost reductions for larger-scale electrolyzer systems, especially by reducing the cost of the corresponding BoP.[25] Developing larger modules requires additional engineering, which governments can support. For example, electrolyzer manufacturer ITM Power received funding from the British government to transform its 0.67 megawatt (MW) stack into a larger-scale 5 MW module, which would serve commercial systems of 100 MW.[26]
Increasing the scale of the manufacturing plant is predicted to produce dramatic cost declines, such as through automation for higher throughput.[27] In the case of ITM Power, the combination of BoP, economies of scale, and reduced labor costs from semi-automated manufacturing are expected to reduce the installed PEM electrolyzer cost to less than $500 per kilowatt. [28]
Nonetheless, there may be tradeoffs between economies of scale at the module and the manufacturing plant. While a small module size may better enable mass manufacturing and standardization, a larger size may achieve higher cost reduction (albeit with a lower learning curve). [29]
The growing scale of announced electrolyzer manufacturing capacity, currently estimated between 9 and 13 gigawatts and expected to grow further, suggests progress toward economies at the manufacturing plant; however, manufacturing output has been slow to materialize, with only around 200 MW installed in 2021 and 2022.[30] Progress will depend on the extent to which existing and future hydrogen policies foster a rapid deployment of renewable hydrogen, enabling an optimal balance between all the parameters discussed here, while also supporting a continuous decline in renewable electricity costs.
CGEP’s Visionary Circle
Corporate Partnerships
Occidental Petroleum Corporation
Tellurian Inc
Foundations and Individual Donors
Anonymous
Anonymous
the bedari collective
Jay Bernstein
Breakthrough Energy LLC
Children’s Investment Fund Foundation (CIFF)
Arjun Murti
Ray Rothrock
Kimberly and Scott Sheffield
Notes
[1] The US is targeting an 80 percent reduction target from a baseline of $5/kg in 2020 to $1/kg in 2030. See US Department of Energy, “U.S. National Clean Hydrogen Strategy and Roadmap,” June 2023, https://www.hydrogen.energy.gov/pdfs/us-national-clean-hydrogen-strategy-roadmap.pdf. This aricle focuses on renewable hydrogen, produced through electrolysis using renewable electricity sources such as wind and solar.
[2] DOE, “US National Clean Hydrogen Strategy and Roadmap.”
[3] Polly Martin, “Green hydrogen | Which type of electrolyser should you use? Alkaline, PEM, solid oxide or the latest tech?,” Hydrogen Insight, July 5, 2023, https://www.hydrogeninsight.com/electrolysers/green-hydrogen-which-type-of-electrolyser-should-you-use-alkaline-pem-solid-oxide-or-the-latest-tech-/2-1-1480577.
[4] Rachel Parkes, “Underestimated? Why Solid-Oxide Electrolysers Could Be the Dark Horse of the Hydrogen Industry,” Hydrogen Insight, May 17, 2023, https://www.hydrogeninsight.com/electrolysers/underestimated-why-solid-oxide-electrolysers-could-be-the-dark-horse-of-the-hydrogen-industry/2-1-1452425.
[5] International Renewable Energy Agency, “Green Hydrogen Cost Reduction: Scaling up Electrolysers to Meet the 1.5⁰C Climate Goal,” 2020. Research organizations may define the electrolyzer systems in different ways; for example, OIES and Fraunhofer use a 4-way split (stack, power electronics, gas conditioning, and BoP).
[6] Ibid; Miller et al., “Green Hydrogen from Anion Exchange Membrane Water Electrolysis: A Review of Recent Developments in Critical Materials and Operating Conditions,” Sustainable Energy Fuels, 2020, 4, 2114–2133, https://doi.org/10.1039/C9SE01240K.
[7] Dmitri Bessarabov, Haijiang Wang, Hui Li, Nana Zhao, PEM Electrolysis for Hydrogen Production: Principles and Applications, CRC Press, Feb 3, 2016, https://books.google.com/books?id=BNuYCgAAQBAJ&dq=%22dryer+losses%22+hydrogen+pem+electrolysis&source=gbs_navlinks_s.
[8] International Energy Agency, “The Future of Hydrogen: Seizing Today’s Opportunities,” June 2019, https://www.iea.org/reports/the-future-of-hydrogen, pg. 44; Marcelo Carmo, David L. Fritz, Juergen Mergel, and Detlef Stolten, “A Comprehensive Review on PEM Water Electrolysis,” International Journal of Hydrogen Energy, Volume 38, Issue 12, April 22, 2013, 4901–4934, https://doi.org/10.1016/j.ijhydene.2013.01.151.
[9] George Bristowe and Andrew Smallbone, “The Key Techno-Economic and Manufacturing Drivers for Reducing the Cost of Power-to-Gas and a Hydrogen-Enabled Energy System,” Hydrogen 2021, 2, 273–300, https://doi.org/10.3390/hydrogen2030015, pg. 282.
[10] A. Hauch et al., “Recent Advances in Solid Oxide Cell Technology for Electrolysis,” Science, Vol. 370, Issue 6513, October 2020, pg. 1, https://www.science.org/doi/10.1126/science.aba6118.
[11] Majid Minary-Jolandan, “Formidable Challenges in Additive Manufacturing of Solid Oxide Electrolyzers (SOECs) and Solid Oxide Fuel Cells (SOFCs) for Electrolytic Hydrogen Economy toward Global Decarbonization,” Ceramics 2022, 5, 761–779. Pg. 762, https://doi.org/10.3390/ceramics5040055; Tom Smolinka et al., “Studie IndWEDe: Industrialisierung der Wasserelektrolyse in Deutschland: Chancen und Herausforderungen für nachhaltigen Wasserstoff für Verkehr, Strom und Wärme,“ Fraunhofer-Institut für Produktionstechnik und Automatisierung IPA, 2018, pg. 43, https://www.ipa.fraunhofer.de/de/Publikationen/studien/studie-indWEDe.html.
[12] Miller et al., “Green Hydrogen from Anion Exchange Membrane Water Electrolysis.”
[13] Aliaksei Patonia and Rahmatallah Poudineh, “Cost-Competitive Green Hydrogen: How to Lower the Cost of Electrolysers,” The Oxford Institute for Energy Studies—OIES Paper: EL 47, January 2022, https://www.oxfordenergy.org/publications/cost-competitive-green-hydrogen-how-to-lower-the-cost-of-electrolysers/; Hans Böhm, Sebastian Goers, and Andreas Zaune, “Estimating Future Costs of Power-to-Gas—A Component-Based Approach for Technological Learning,” International Journal of Hydrogen Energy, 44, 2019, 30789-30805 https://doi.org/10.1016/j.ijhydene.2019.09.230; Tom Smolinka et al., “Studie IndWEDe: Industrialisierung der Wasserelektrolyse in Deutschland: Chancen und Herausforderungen für nachhaltigen Wasserstoff für Verkehr, Strom und Wärme,“ Fraunhofer-Institut für Produktionstechnik und Automatisierung IPA, 2018, https://www.ipa.fraunhofer.de/de/Publikationen/studien/studie-indWEDe.html.
[14] IEA, “The Future of Hydrogen.”
[15] Full load hours refers to hours when an asset is utilized at its maximum installed capacity. At full load the electrolyzer runs on electricity so as to produce the greatest quantity of hydrogen possible. Especially for renewable hydrogen production—which depends on the availability of renewable electricity—electrolyzers may not always run on full load.
[16] IEA, “The Future of Hydrogen.”
[17] This refers to how much hydrogen is produced given a certain electrical input. One needs to be very careful to note whether the efficiencies (in percentage terms or in kWh of electricity per kg of hydrogen) refer to the stack or the system. In studies, the first one usually refers to the stack; studies usually specify whether the second refers to the stack or the system or specify both (IRENA).
[18] Aurora Energy Research, “EP. 128 Nils Aldag, Co-Founder and CEO, Sunfire,” Energy Unplugged, https://soundcloud.com/user-564729441/aurora-energy-unplugged-nils-aldag.
[19] Current density (in Ampere per square centimeter) refers to the “amount of charge flowing through a specific cross-sectional area.” In the case of hydrogen production, the amount of hydrogen produced is proportional to the current density.
[20] IRENA, “Green Hydrogen Cost Reduction: Scaling up Electrolysers to Meet the 1.5⁰C Climate Goal,” pg. 43–44.
[21] Haider Ali Khan et al., “Designing Optimal Integrated Electricity Supply Configurations for Renewable Hydrogen Generation in Australia,” iScience, 2021 Jun 25; 24(6): 102539. https://doi.org/10.1016/j.isci.2021.102539.
[22] IEA, “The Future of Hydrogen,” pg. 46, https://www.iea.org/reports/the-future-of-hydrogen; Patonia and Poudineh, “Cost-Competitive Green Hydrogen.”
[23] IRENA, “Green Hydrogen Cost Reduction,” pg. 33;
Patonia and Poudineh, “Cost-Competitive Green Hydrogen,” pg. 24.
[24] Patonia and Poudineh, “Cost-Competitive Green Hydrogen.”
[25] IRENA, “Green Hydrogen Cost Reduction,” pg. 71.
[26] ElementEnergy, “Gigastack: Bulk Supply of Renewable Hydrogen,” Element Energy Limited, February 2020, https://gigastack.co.uk/content/uploads/2020/06/Gigastack-Phase-1-Public-Summary.pdf.
[27] IRENA, “Green Hydrogen Cost Reduction.”
[28] ElementEnergy, “Gigastack: Bulk Supply of Renewable Hydrogen,” Element Eneryg Limited, February 2020.
[29] IRENA, “Green Hydrogen Cost Reduction.”
[30] Hydrogen Council, “Hydrogen Insights 2023,” May 2023, https://hydrogencouncil.com/en/hydrogen-insights-2023/; IEA, “Electrolysers,” accessed in June 2023, https://www.iea.org/reports/electrolysers.
More on Energy Explained Energy Explained
Assessing the Energy Impacts of the One Big Beautiful Bill Act
This special CGEP blog series, featuring six contributions from CGEP scholars, analyzes the potential impacts of the OBBBA across a range of sectors.

The US has a Hydrogen Demand Problem – Despite Generous Incentives
When the Inflation Reduction Act (IRA) was passed in August 2022, it triggered unprecedented enthusiasm among potential hydrogen suppliers.

Why China’s Renewable Ammonia Market Is Poised for Significant Growth
China’s commitment to what it calls its “dual carbon” goals of carbon neutrality by 2060 and to ammonia’s potential role as a hydrogen derivative and carrier have fostered expectations that its renewable ammonia market will expand significantly and thus so will production.
How Countries Are Planning to Produce Hydrogen
This blog post is a comparative analysis of the various production pathways each nation is considering.

Relevant
Publications
La Filière Hydrogène dans le Contexte Géopolitique Mondial
La géopolitique de l’hydrogène s’inscrit dans un nouvel ordre énergétique mondial opposant les pétro-États, fondés sur l’exportation d’hydrocarbures, et les […]

Trump’s One Big Beautiful Bill Act Has Given the US Hydrogen Industry Certainty — but Where Will the Green Energy Come From?
H2 projects will have to compete for a shrinking pipeline of zero-carbon electricity with energy-intensive data centres.



